Is So3 Ionic Or Covalent

CaF2 is an ionic compound because when the metal combines with nonmetal, it commonly forms an ionic compound. Here, Ca is a metal and F is a nonmetal. And then when they combine, it forms an ionic compound.
Well, now you have got to know that CaF2 is an ionic compound, but permit me explicate the in-depth reason why CaF2 is an ionic chemical compound.
Why is CaF2 an ionic compound?
As mentioned in a higher place, you lot tin can only remember that when the metal combines with nonmetal, the bond between them is an ionic bond.
Here in CaF2, the Ca atom is a metal and the F atom is a nonmetal.
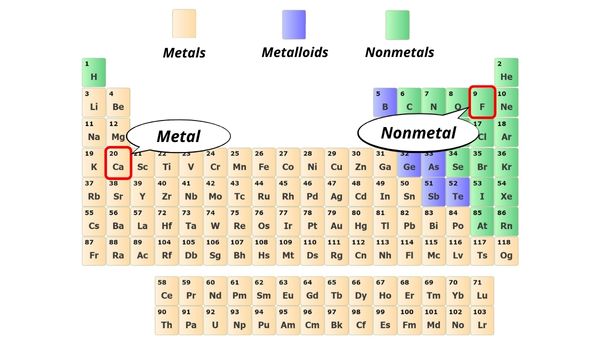
Hence the bail betwixt them is an ionic bond.
How does the ionic bond form betwixt Ca and F?
In CaF2, at that place are two atoms;
Ca and F.
Near Calcium (Ca):
Calcium atom have 20 electrons.
The electrons arrangement in Calcium (Ca) is two, 8, 8, 2.
So the outermost orbit of Calcium atom has two electrons.
At present Calcium is a metallic and the metals are highly electropositive (that means they have the tendency to lose electrons and become positive ions.)
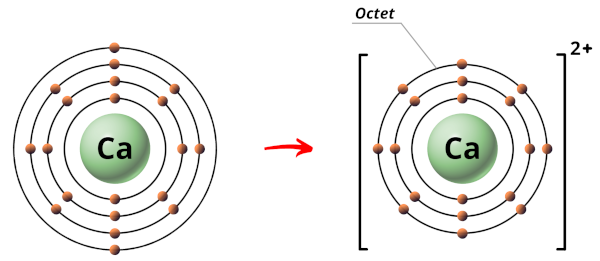
Hence during the chemical reaction, the Calcium cantlet will lose ii electrons to form a stable octet.
Most Fluorine (F):
Fluorine atom have 9 electrons.
The electrons arrangement in Fluorine (F) is 2, 7.
So the outermost orbit of Fluorine atom has 7 electrons.
At present Fluorine is a nonmetal and the nonmetals are highly electronegative (that means they accept the tendency to gain electrons and become negative ions.)
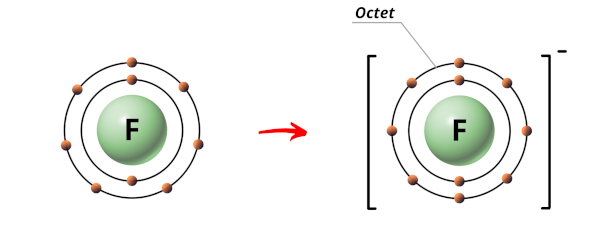
Hence during the chemic reaction, the Fluorine cantlet will proceeds one electron to class a stable octet.
What happens when Ca and F combine?
When Ca and F combine with each other, the electron transfer takes place from Calcium atom to Fluorine atom (i.east from Ca to F).
In other words, the Calcium cantlet (Ca) loses 2 electrons and the Fluorine atoms (F) gains 1-1 electrons.
Due to this, the Calcium becomes a positive ion (Caii+) and Fluorine becomes a negative ion (F–).
Now considering of the positive charge of Calcium ion and negative accuse of Fluorine ion, the electrostatic force of attraction is produced betwixt them.
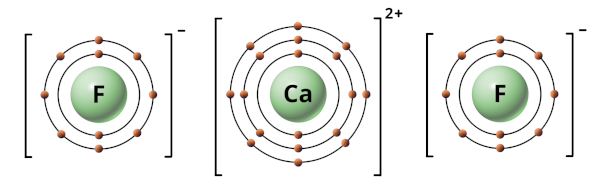
This electrostatic force between Calcium ion and Fluorine ions results in an ionic bond between them.
Hence, CaF2 is an ionic compound.
I hope y'all have understood the reason why CaF2 is an ionic compound.
Check out other compounds to encounter whether they are ionic or covalent;
Is K2O Ionic or Covalent?
Is KBr Ionic or Covalent?
Is KI Ionic or Covalent?
Is CaCO3 Ionic or Covalent?
Is MgBr2 Ionic or Covalent?
Is So3 Ionic Or Covalent,
Source: https://knordslearning.com/is-caf2-ionic-or-covalent/
Posted by: ocasiogrou1983.blogspot.com


0 Response to "Is So3 Ionic Or Covalent"
Post a Comment