Electron Configuration For O 2
Electron Configurations
The content that follows is the substance of General Chemistry Lecture 26. In this lecture nosotros continue the discussion of Quantum Numbers and their use in Electron Configurations too every bit the human relationship of electron configuration to the periodic properties of the elements.
Electron Configuration
Electron configurations are the summary of where the electrons are effectually a nucleus. As we learned earlier, each neutral atom has a number of electrons equal to its number of protons. What nosotros will do now is identify those electrons into an arrangement around the nucleus that indicates their free energy and the shape of the orbital in which they are located. Here is a summary of the types of orbitals and how many electrons each can incorporate:
And then based on what nosotros know near the quantum numbers and using the chart above, you need 2 electrons to fill an s orbital, 6 electrons to make full a p orbital, ten electrons to fill a d orbital and 14 electrons to fill the f orbital. BUT what we haven't discussed is how these orbitals get filled...the order of make full.
Social club of Fill
The social club in which electrons are placed into the orbitals is based on the society of their energy. This is referred to every bit the Aufbau principle. The lowest free energy orbitals fill up first. Just similar the quantum numbers themselves this club was determined by calculation and is summarized by the following chart:
or you tin just employ the periodic table:

How to Write an Electron Configuration
The symbols used for writing the electron configuration starting time with the shell number (north) followed by the type of orbital and finally the superscript indicates how many electrons are in the orbital.
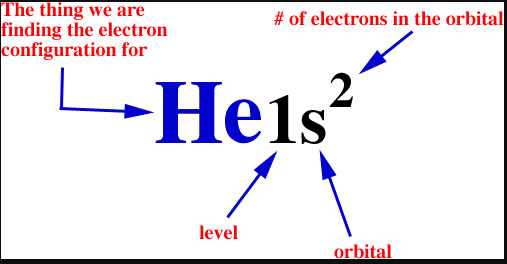
For example:
Looking at the periodic table, you tin can see that Oxygen has 8 electrons. Based on the order of fill up to a higher place, these 8 electrons would fill in the following order 1s, 2s so 2p. And so Oxygen'due south electron configuration would be O 1s22s22pfour .
Special Cases
Configurations of ions present a special case of electron configuration and also demonstrate the reason for the formation of those ions in the first place.
If you need to write the full electron configuration for an anion, so you are just adding additional electrons and the configuration is simply connected.
For case, we know that Oxygen ever forms 2- ions when it makes an ion. This would add 2 electrons to its normal configuration making the new configuration: O2- 1s22stwo2pvi . With 10 electrons you should note that oxygen's electron configuration is now exactly the aforementioned equally Neon's. Nosotros talked well-nigh the fact that ions form because they can become more stable with the gain or loss of electrons to become similar the noble gases and now y'all tin can actually encounter how they become the same.
The electron configurations for Cations are too made based on the number of electrons only at that place is a slight departure in the way they are configured. First yous should write their normal electron configuration and then when yous remove electrons you accept to take them from the outermost crush. Note that this is non always the aforementioned fashion they were added.
Hither is an case of what I hateful:
Iron has 26 electrons so its normal electron configuration would be: Fe 1s22s22psix3s23p64s23dsix
When nosotros brand a 3+ ion for Iron, we need to take the electrons from the outermost shell get-go then that would be the 4s shell Non the 3d trounce: Atomic number 26iii+ 1stwo2sii2p63s23pvi3dv
Ane other note on writing electron configurations: A short cut. When writing some of the lower table configurations the total configuration can be fairly long. In these cases, you tin use the previous noble gas to abridge the configuration as shown below. You just accept to end the configuration from where the element of group 0 leaves it:

Exceptions
As with every other topic nosotros take covered to engagement there are exceptions to the gild of fill too. But based on the electron configurations that are generated, these exceptions are easy to empathize.
In the d block, specifically the groups containing Chromium and Copper, there is an exception in how they are filled.
Here are the actual configurations:
In these columns, the 4s and 3d
Practice, Exercise, Practice
There are lots of quizzes on electron configurations you can practise with located here
Orbital Diagrams
Some other mode to represent the social club of fill for an atom is by using an orbital diagram often referred to as "the lilliputian boxes":

The boxes are used to represent the orbitals and to show the electrons placed in them. The lodge of make full is the aforementioned merely every bit you can run across from in a higher place the electrons are placed singly into the boxes before filling them with both electrons. This is called Hund's Rule: "Half fill up before you lot Full make full" and once again this rule was established based on energy calculations that indicated that this was the way atoms actually distributed their electrons into the orbitals.
Periodic Properties
One of the really cool things nearly electron configurations is their relationship to the periodic tabular array. Basically the periodic table was constructed and so that elements with similar electron configurations would exist aligned into the same groups (columns).

Periodic Table showing last orbital filled for each element
The periodic tabular array shown above demonstrates how the configuration of each element was aligned then that the concluding orbital filled is the same except for the beat. The reason this was done is that the configuration of an element gives the element its properties and similar configurations yield like properties.
Let's go through some of the Periodic Properties that are influenced directly by the electron configuration:
Diminutive Size
| The size of atoms increases going down in the periodic table. This should exist intuitive since with each row of the table you lot are adding a shell (northward). What is non as intuitive is why the size decreases from left to right. Simply again the construction of the electron configuration gives usa the answer. What are you lot doing as y'all become beyond the periodic table? Answer, adding protons to the nucleus and adding electrons to the valence crush of the element. What is not changing as you cross a menstruum? Respond, the inner shell electrons. Then think of information technology this manner, the inner vanquish electrons are a shield against the pull of the nucleus. As you cross a period and increase the number of protons in the nucleus you increase its pull just since you are only adding electrons to the new crush the shield is not increasing but remains the same all the mode across. This means the pull on the electrons existence added to the valence shell is increasing steadily all the mode beyond. What happens if yous pull harder on the electrons? Well, they come up closer to the nucleus and the size of the atom decreases. The issue of the nucleus pulling on the electrons existence added across a period is called the effective nuclear charge and is calculated as ZEff = #protons - Cadre # Electrons. So for example the pull felt by Sulfur would exist ZEff = 16 - x = +6 | 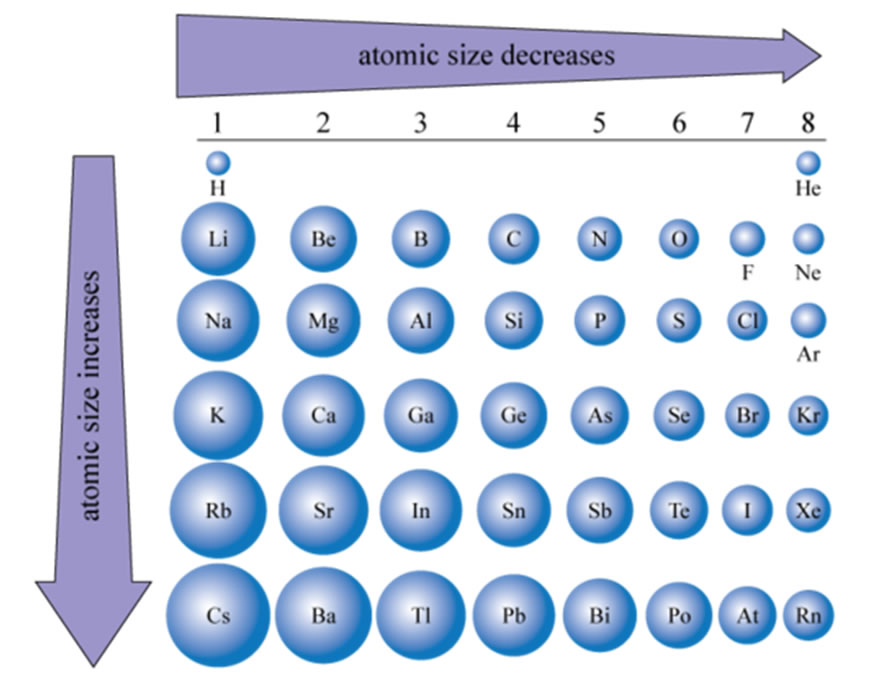 |
Electronegativity
Electronegativity may be the virtually important of the periodic properties you tin larn and understand since so many other backdrop are depend on its value. Electronegativity is an atoms ability to pull electrons towards itself.
Electronegativity is generally expressed past the Pauling Calibration and the values were determined experimentally. The table below shows the scale values for the elements.
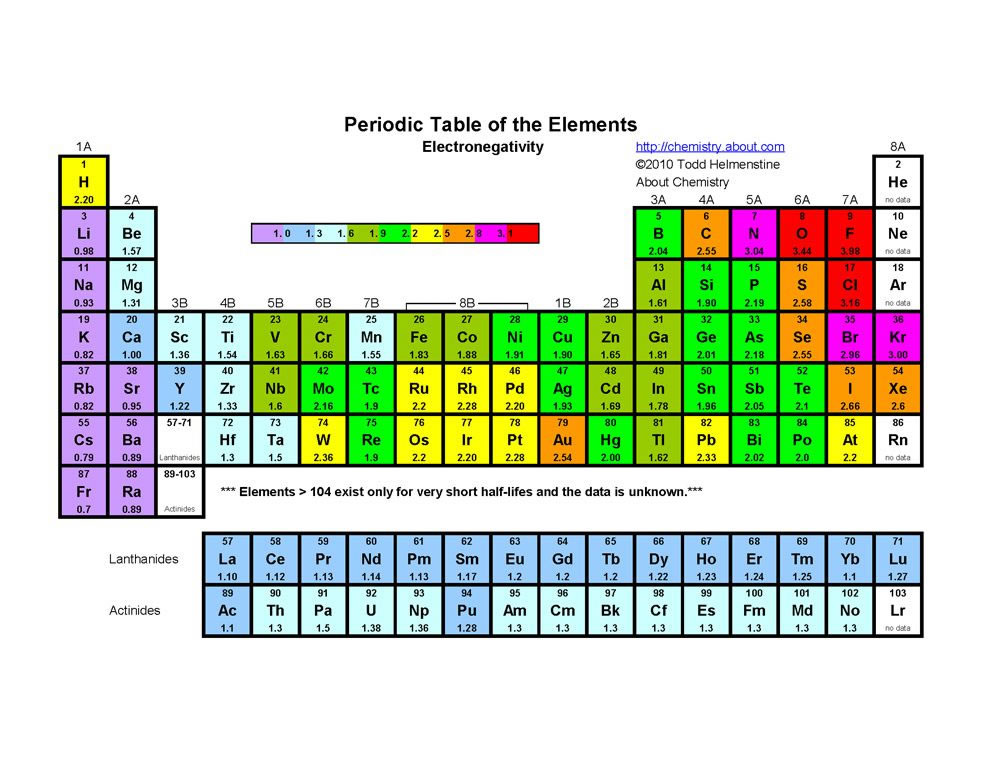
The electronegativity values increase from left to correct and bottom to top in the periodic tabular array excluding the Noble gases. The well-nigh electronegative element is Fluorine.
From these electronegativity values we tin derive the patterns of two other periodic properties: Ionization Energy and Electron Affinity.
| | Ionization Free energyIonization free energy is the amount of free energy required to remove an electron from an atom. All ionization energies are positive values because all of these removals (even those for elements that grade positive ions) require input of free energy. The more electronegative the element, the higher the ionization eneregy. |
Electron AffinityThe Electron Affinity of an element is the amount of energy gained or released with the addition of an electron. The electronegativity and Electron Analogousness increases in the same pattern in the periodic table. Left to correct and bottom to top. | |
Electron Configuration For O 2,
Source: https://www.chem.fsu.edu/chemlab/chm1045/e_config.html
Posted by: ocasiogrou1983.blogspot.com



0 Response to "Electron Configuration For O 2"
Post a Comment